CSSD Cleaning Monitoring: Safety & Traceability
Why CSSD Cleaning Monitoring Is Critical
Cleaning is the first and most important step in reprocessing. If organic residues remain on instruments, sterilization may fail regardless of the cycle parameters.
A proper CSSD cleaning monitoring strategy must control all critical variables involved in the washing cycle:
- Time
- Temperature
- Detergent type and concentration
- Water quality
- Mechanical action
Given the complexity of these parameters, relying on manual checks is no longer enough. Automated monitoring ensures consistency, repeatability, and compliance.
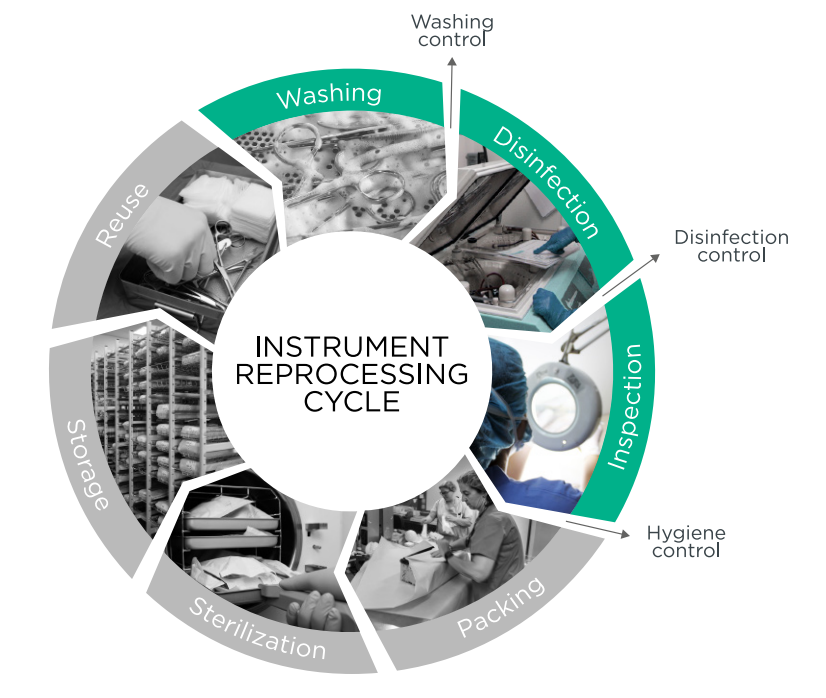
A diagram illustrating the seven-stage sterile processing cycle, emphasizing critical control points for washing, disinfection, and inspection
Challenges in Cleaning Validation
Many CSSDs still face key limitations when validating cleaning processes:
- No verification of every load and rack
- Lack of objective and standardized evaluation
- Limited or no traceability
- Use of ATP-based systems that do not detect proteins or prions
Protein residues are among the most critical contaminants in medical device reprocessing. Unlike ATP systems, protein-based monitoring solutions allow a more accurate evaluation of cleaning effectiveness—including risks associated with prions.
Advanced CSSD Cleaning Monitoring with Terragene Solutions
A complete CSSD cleaning monitoring approach integrates multiple technologies to ensure performance, hygiene control, and full traceability.
For cleaning performance monitoring, Chemdye® Splat indicators use a specially formulated test soil that simulates organic residues typically found on medical instruments. These highly versatile indicators can be used in both washer-disinfectors and ultrasonic washers, depending on the holder selected, and can be processed with the load or in an empty chamber. They are suitable for both routine monitoring and equipment performance qualification.
For ultrasonic cleaning processes, CDWU & CDWU-Z cavitation indicators combined with holders allow you to evaluate cavitation energy distribution and detect ineffective zones inside the washer. When paired with CAVITEST® auto-reader, results become fully objective, delivering quantifiable data and clear Pass/Fail outcomes.
Cavitest and CDWU-Z – Quick User Guide
After cleaning, surgical instruments must be disinfected to ensure safe subsequent handling and processing. Thermal disinfection with moist heat, is the most common method for disinfection of medical devices in the hospital setting. Thermodisinfection can be specifically monitored using IT27W indicators,
For hygiene monitoring, Terragene introduces advanced protein detection systems such as PRO1 MICRO & PRO1 ENDO, enabling quantitative measurement of protein residues on surfaces and inside cannulated instruments. Together with the MiniPro auto-reader, these systems enable incubation and quantitative readout of protein detection pens such as Chemdye® PRO1 MICRO, designed for the detection of surface proteins and allergens.
PRO1 Micro Hygiene Monitoring System and Bionova MiniPro auto-reader for the detection and quantification of proteins on surfaces.
Finally, all data can be integrated into Bionova® Q CSSD software, enabling real-time monitoring, full traceability, and complete visibility of the reprocessing cycle—from cleaning to sterilization and final release.
Upgrade your CSSD cleaning monitoring strategy with real, measurable results.
Join our webinar:
“Ensuring Effective Cleaning: Advanced Monitoring Solutions for CSSDs”
Discover how to implement automated monitoring, improve traceability, and ensure cleaning effectiveness in every cycle.
Choose the session that best fits your schedule:
🌍 Region 1: Europe • Middle East • Asia (New Delhi, Bangkok / Hanoi & Jakarta) → Register Now
🌏 Region 2: Asia (Hong Kong, Seoul, Tokyo, Shanghai, Beijing) • Australia • North America → Register Now
Save your spot for the next session:
North America • Australia • NZ
East Asia (Seoul, Tokyo, Shanghai, Beijing)


